- Blog
- Gta real life meme
- Jonny walker ktm bike
- Download microsoft flight simulator x gold edition free
- Kensington docking sattion no clone mode
- Dcuo sins of the father
- Where is the quick analysis tool in excel for mac-
- Ea cricket 2007 world cup edition
- Teraterm logging
- Stata mp serial number lic file
- Fda iso 13485
- Boy commandos 1
- Mac os high sierra dmg download direct
- Undertaker wrestlemania 22 full match
- Shaurya movie dialogue
- Where to download hd music videos for free
- Crystal report viewer in visual studio 2012
- Outlook express 6 xp
- Crack tkaraoke pro using ollydbg
The move forms part of a larger push for global regulatory harmonization, including via the Medical Device Single Audit Program.
#Fda iso 13485 iso
The proposed FDA rule for harmonizing medical device quality management systems with ISO 13485 is “an important next step,” an International Organization for Standardization (ISO) committee said in a post Monday, August 27. The FDA seeks to blend the third iteration of the international consensus standard for quality management systems with 21 CFR 820 as part of an effort to “reduce compliance and recordkeeping burdens” related to quality system regulation requirements. The revisions will also modernize the regulation”. The revisions are intended to reduce compliance and recordkeeping burdens on device manufacturers by harmonizing domestic and international requirements.

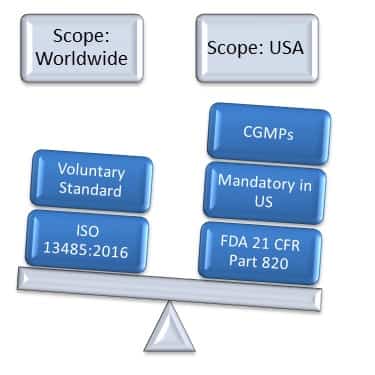
The revisions will supplant the existing requirements with the specifications of an international consensus standard for the medical device manufacturer, ISO 13485:2016. “FDA intends to harmonize and modernize the Quality System regulation for medical devices.

regulatory requirements.”įDA then published a rule in Spring 2018, stating: In the spirit of global harmonization of quality management systems, the FDA is considering an evaluation/mapping of the 13485 clauses to the appropriate U.S. “With the publishing of the 13485:2016 revision, there has been industry evaluation of the regulatory requirement linkages to the standard. Late in February 2018, the FDA announced that as part of global harmonization efforts around quality management systems, the agency is considering adding clauses from ISO 13485:2016 to the appropriate US medical device quality regulatory requirements.įDA press officer Stephanie Caccomo stated via email: In addition, photographic documentation is provided to further support evidence noted.Ĭontact us to review an example report, or further discuss how we meet the growing needs of the medical device industry.ISO Prepares for FDA’s Proposed Overhaul of Medical Device Quality System Regulation
#Fda iso 13485 pro
Pro QC performs FDA 21 CFR Part 820 audits that incorporate an evaluation of each of the following regulation components: The dates and results of quality audits and reaudits shall be documented.” A report of the results of each quality audit, and reaudit(s) where taken, shall be made and such reports shall be reviewed by management having responsibility for the matters audited. Corrective action(s), including a reaudit of deficient matters, shall be taken when necessary. Quality audits shall be conducted by individuals who do not have direct responsibility for the matters being audited. “Each manufacturer shall establish procedures for quality audits and conduct such audits to assure that the quality system is in compliance with the established quality system requirements and to determine the effectiveness of the quality system.
In Section 820.22 of the Code, a quality audit is prescribed.
#Fda iso 13485 code
Rather, the regulation provides the framework that all manufacturers must follow by requiring that manufacturers develop and follow procedures and fill in the details that are appropriate to a given device according to the current state-of-the-art manufacturing for that specific device.”ĬFR – Code of Federal Regulations Title 21 “The QS regulation applies to finished device manufacturers who intend to commercially distribute medical devices. A finished device is defined in 21 CFR 820.3(l) as any device or accessory to any device that is suitable for use or capable of functioning, whether or not it is packaged, labeled, or sterilized.īecause the regulation must apply to so many different types of devices, the regulation does not prescribe in detail how a manufacturer must produce a specific device. FDA’s Quality System Regulation Part 820 is harmonized with ISO 13485:1996, which is based on ISO 9001:1994. Food and Drug Administration regulates current good manufacturing processes (CGMP’s) also known as FDA 21 CFR Part 820. To ensure medical devices are safe and effective, the U.S. is the relationship to FDA 21 CFR Part 820. A common question we receive in relation to regulatory requirements in the U.S. Our recent newsletter Feature Article highlighted ISO 13485 audits applicable to the medical device industry.